Let’s re-examine the maximal entropy principle ( an earlier post) from a probablistic point of view (se the post on ML/AI-probability-I for use of generating functions to count states), starting with
![Rendered by QuickLaTeX.com \[\Omega(N,V,E)=\sum'_\mu1\quad \mbox{$\Omega=$ sum over the microstates}\]](http://abadonna.physics.wisc.edu/wp-content/ql-cache/quicklatex.com-965d54239079ed5cb8aa98ea5acc3399_l3.png)
where we sum over identifiers of microstates that result in the same macrostate. Divide
![Rendered by QuickLaTeX.com \[1=\sum'_\mu {1\over \Omega(N,V,E)}\]](http://abadonna.physics.wisc.edu/wp-content/ql-cache/quicklatex.com-ca1055c361f23a9f162bcd9ebac819df_l3.png)
which we interpret as stating that the probability that you reach into an ensemble of systems in macrostate ![]() and pull out a system whose microstate is
and pull out a system whose microstate is ![]() .
.
![]()
The maximal entropy principle is a maximal probability principle, the most likely macrostate is the equilibrium macrostate. We maximize the probability that the entire universe is in a given macrostate (![]() )
)
![]()
![]()
Restrict our attention to energy only, and we suppose that ![]()
![]()
And so a generating function is born! The un-normalized probabilities we write as
![]()
The probability that ![]() has
has ![]() is
is
![]()
and the equilibrium partition of energy is that which maximizes the probability
![]()
which brings us back to the First law, compatibility requires ![]() .
.
Multiply this equation by ![]() and use the first law; when
and use the first law; when ![]() is the equilibrium energy of
is the equilibrium energy of ![]() ;
;
![]()
which we recognize is the ![]() -theorem (since we have held
-theorem (since we have held ![]() constant)
constant)
![]()
is the equilibrium condition for constant ![]() and constant
and constant ![]() .
.
In summary, here is the interpretation; create a new generating function by introducing an intensive variable ![]() . It is intensive because it has nothing to do with
. It is intensive because it has nothing to do with ![]() , it has everything to do with
, it has everything to do with ![]() . The new generating function is a re-expression of the summed probabilities that
. The new generating function is a re-expression of the summed probabilities that ![]() is in a certain macrostate when in contact with
is in a certain macrostate when in contact with ![]() . Maximizing that probability will give you equilibrium values of extensive properties of
. Maximizing that probability will give you equilibrium values of extensive properties of ![]() .
.
While we are on this subject let’s show that the transformation from one thermodynamic potential
![]()
by Legendre transformation
![]()
is paralleled in statistical mechanics by Laplace transformation of partition/generating functions. Starting with the generating function ![]() ,
, ![]() we have seen that
we have seen that
![]()
![]()
and in the thermodynamic limit
![]()
in which ![]() computed at the most probable (equilibrium) energy of the system in equilibrium with the reservoir.
computed at the most probable (equilibrium) energy of the system in equilibrium with the reservoir.
Let’s quickly review the transformation at the potential/thermodynamic level. Start with the definition of extensive energy
![]()
Differentiate with respect to ![]() and set
and set ![]()
![]()
![]()
Use the first law
![]()
![]()
to arrive at the Gibbs-Duhem relation
![]()
Let
![]()
![]()
and finally introduce the grand potential
![]()
for which
![]()
![]()
Let’s create another Laplace transform of ![]()
![]()
Calling ![]() we see that this can be used as a generating function counting variable in the absence of energetics. Think of this as
we see that this can be used as a generating function counting variable in the absence of energetics. Think of this as
![]()
and expand around the equilibrium system occupancy ![]() holding
holding ![]() fixed
fixed
![]()
![]()
![]()
To evaluate ![]() use Gibbs-Duhem
use Gibbs-Duhem
![]()
![]()
![]()
and the triple-product identity, and the isothermal compressibility ![]()
![]()
![]()
![]()
![Rendered by QuickLaTeX.com \[Q[T,V,\mu]=\int e^{-{1\over k_BT}\Big(F_*-\mu N_*+{V(N-N_*)^2\over 2N_*^2k_T}+\cdots\Big)} \, dN\]](http://abadonna.physics.wisc.edu/wp-content/ql-cache/quicklatex.com-20e8bf2b5f5a8967961dc6457be3fb50_l3.png)
![]()
Integrate, take logs, remember ![]() and
and ![]() and
and ![]() ,
, ![]() are intensive, and
are intensive, and ![]() , so in the thermodynamic limit
, so in the thermodynamic limit
![]()
![]()
Example. Consider our gas of ![]() particles in a box of volume
particles in a box of volume ![]() for which
for which ![]() . Then
. Then
![]()
(in which you recognize the generating function if we call ![]() ). The results for the average system occupancy when in chemical/matter equilibrium with a particle reservoir looks very Bose-Einsteinish
). The results for the average system occupancy when in chemical/matter equilibrium with a particle reservoir looks very Bose-Einsteinish
![]()
Example ![]() for noninteracting lattice gases of
for noninteracting lattice gases of ![]() cells can be constructed from the requirement that
cells can be constructed from the requirement that ![]() is extensive, the entire gas is simply
is extensive, the entire gas is simply ![]() copies of a single cell lattice gas, and the cells are distinguishable by position in the lattice, so
copies of a single cell lattice gas, and the cells are distinguishable by position in the lattice, so ![]() where
where ![]() is the grand canonical partition function of a single cell, therefore
is the grand canonical partition function of a single cell, therefore ![]() . For a single cell with occupancies
. For a single cell with occupancies ![]() by identical particles there is one way to put
by identical particles there is one way to put ![]() particles in a cell, so
particles in a cell, so ![]() where
where ![]() is the energy of the cell containing
is the energy of the cell containing ![]() particles. For example if
particles. For example if ![]() are the allowed occupances and there is a binding energy
are the allowed occupances and there is a binding energy ![]() then
then ![]() and
and ![]() for the entire system. The equation of state is gotten by eliminating
for the entire system. The equation of state is gotten by eliminating ![]() between
between
![]()
In thermodynamics we obtain everything from a potential and the first law. In statistical physics we obtain everything from a partition (generating) function correlated with a potential.
Sometimes the variables that appear in ![]() may not be the most convenient for some types of calculations, suppose that we need to use
may not be the most convenient for some types of calculations, suppose that we need to use ![]() ,
, ![]() , and
, and ![]() as the independent variables? We transform into this set using Legendre transformations in classical thermodynamics. For example we know that
as the independent variables? We transform into this set using Legendre transformations in classical thermodynamics. For example we know that
![]()
we could convert to a new potential\index{Legendre transformation}
![]()
differentiate
![]()
![]()
How do we accomplish the same transformation in the statistical mechanics context?
We do it with Laplace transformation to a new generating function
![]()
Let’s demonstrate again that this is equivalent to the transformation
![]()
To do this expand ![]() about the most probable volume for the system (hold
about the most probable volume for the system (hold ![]() fixed)
fixed)
(1) 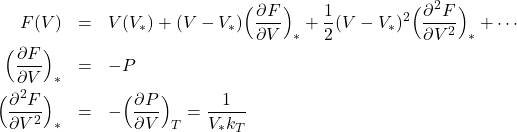
this allows us to write
(2) 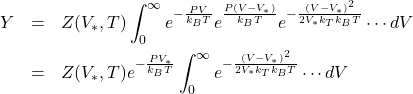
take logs of both sides and multiply by ![]() ;
;
![]()
![]()
the quantity ![]() is of the same order as
is of the same order as ![]() , but the integral is Gaussian and is of the order of
, but the integral is Gaussian and is of the order of ![]() , and
, and ![]() , so in the thermodynamic limit we obtain an extensive function
, so in the thermodynamic limit we obtain an extensive function ![]() .
.
![]()
Therefore we learn that the process of hopping from one thermodynamic potential in thermodynamics is implemented at the statistical mechanical level by Laplace transforming one partition function to get a new one.
Notice that we have been able to write all of the state distribution functions as normal distribution functions with variances given by thermodynamical quantities.